Consensus™
Consensus Target Design for viruses, bacteria, fungi, and parasites.
Providing completely automated design of a PCR assay with unprecedented coverage, sensitivity and specificity across variations of a target.
Design a simple assay to detect and differentiate between closely related genomes.
Consensus ™-has been rigorously validated for the detection of thousands of viruses, bacteria and human targets.
It is useful for applications such as: infectious disease variants for bacteria, viruses, human genomic targets, and mRNA profiling. It also provides consensus design for single panels (i.e. detecting all variants of pandemic 2009 influenza A virus or one set of “universal” primers for all 10,000 FluA variants).

The Challenges of “Freeware”
We often see researchers relying on outdated, antiquated tools. If assay design is mission critical to your organization, why would you want to use a sub-optimal tool?
Common challenges in multiplex panel design include a lengthy and costly design cycle. This can often be the result of both a knowledge gap within the organization along with freeware and sub-optimal design tools.
Consensus™ solves these problems by addressing the following:
Best-in-class Design Algorithms
- The novel assay design algorithm behind Consensus™ is built on a set of empirically-derived thermodynamic parameters that calculate oligo hybridizations from first principles (rather than derivative complementarity rules).
- A wide variety of potential PCR artifacts are accounted for in Consensus™ such as: false negatives caused by target secondary structure, undesired primer-primer interactions, false primer-amplicon interactions, false amplicons to background genomes, and low coverage due to sequence variations.
- Consensus™ can handle large playlists such as the entire human genome, human RefSeq, human microbiome, bacterial diversity panel, soil microbes, or all known viruses, with many pre-imported sequences provided in the software for common use.
Fewer Laboratory Development Cycles
- Consensus™ designs tend to work out of the box, reducing the number of lab iterations needed to arrive at a viable assay. This saves time (3-8 months on average) and associated costs, and gets your assay out faster.
Scalable Designs
- Custom platform, laboratory, and business requirements (e.g. multiplexing) can be accommodated by Consensus™ to optimize designs for use in high volume commercial laboratories, reducing costs of running your assay at scale.

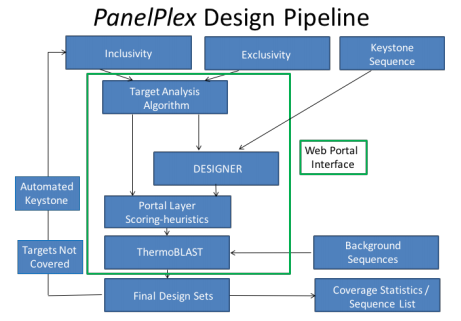
Consensus Packages
Base Package
With a base Consensus license, each licensed user (seat) can:
- Access Consensus software through a secure WebUI portal.
- Design singleplex PCR primers/probes optimized to assay a target of homologous sequences.
- Include assay background genomes to build designs that avoid unwanted amplifications.
- Add and account for fluorophores/quenchers/tails in assay design oligos.
- Review comprehensive PCR assay design results.
- Execute up to 2 concurrent PCR assay design jobs.
- Execute up to 100 hrs of hosted hybridization analysis with unlimited results storage.
- Share target sequences and analysis results with other users.
Interested in learning more about Consensus™?
Consensus™ regularly saves organizations 6+ months in work by using inclusivity and exclusivity panels for diagnostic quality assays.
See how Consensus™ can benefit your organization.

