Amplification Efficiency for primers Is Not Exponential
Myth 6: At the End of PCR, Amplification Efficiency Is Not Exponential Because the Primers or NTPs Are Exhausted or the Polymerase Looses Activity
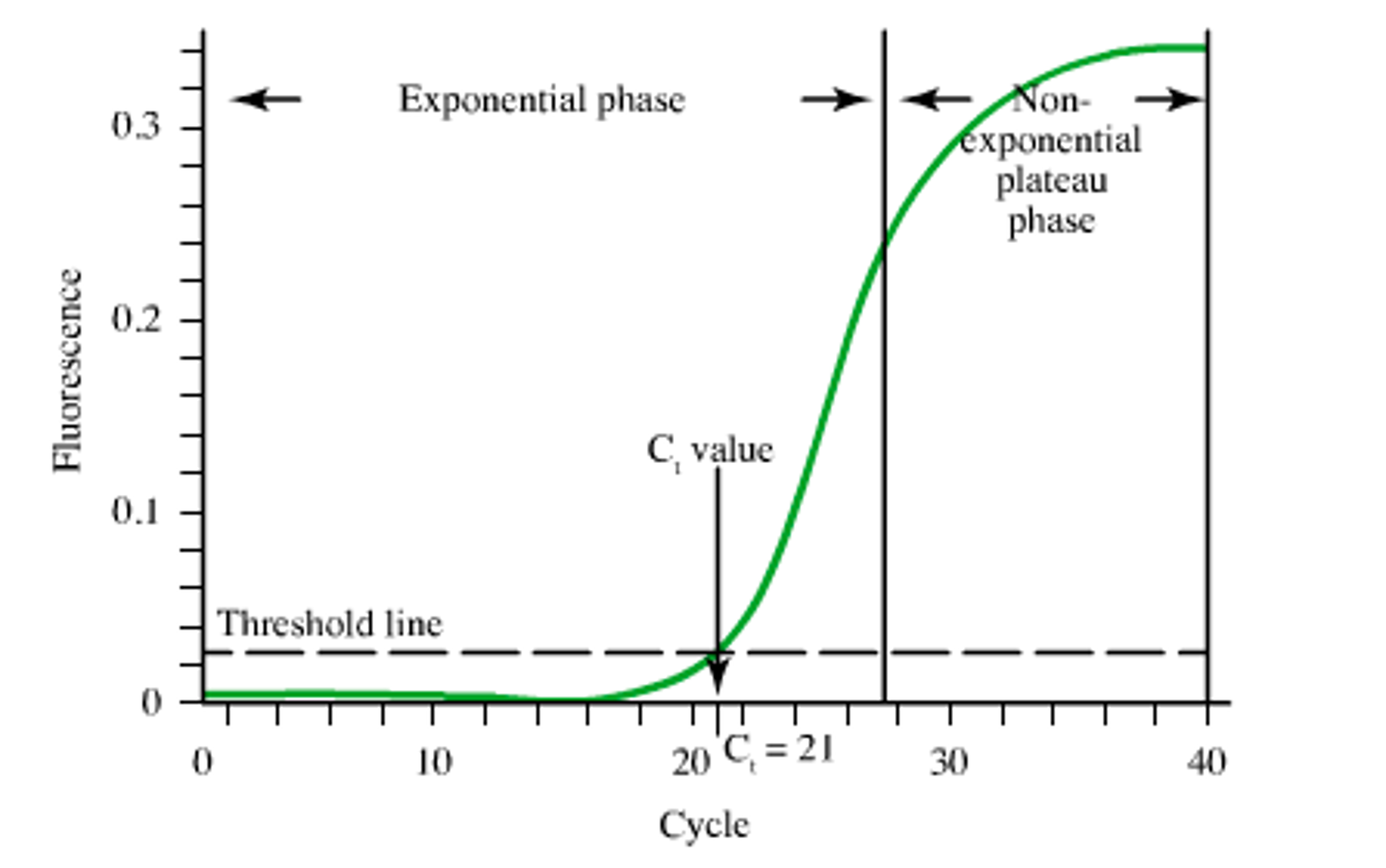 PCR amplification occurs with a characteristic “S” shape. During the early cycles of PCR, the amplification is exponential. During the later stages of PCR, saturation behavior is observed, and the amplification efficiency of PCR decreases with each successive cycle. What is the physical origin of the saturation and why is the explanation important for PCR design? Most practitioners of PCR believe that saturation is observed because either the primers or the NTPs are exhausted or the polymerase looses activity.
PCR amplification occurs with a characteristic “S” shape. During the early cycles of PCR, the amplification is exponential. During the later stages of PCR, saturation behavior is observed, and the amplification efficiency of PCR decreases with each successive cycle. What is the physical origin of the saturation and why is the explanation important for PCR design? Most practitioners of PCR believe that saturation is observed because either the primers or the NTPs are exhausted or the polymerase looses activity.
The idea of lost polymerase activity is historical. In the early days of PCR, polymerase enzymes did loose activity with numerous cycles of PCR. Modern thermostable engineered polymerases, however, are quite robust and exhibit nearly full activity at the end of a typical PCR. The idea of one or more of the NTPs or primers being limiting reagents is perfectly logical and consistent with chemical principles but is not correct for the concentrations that are usually used in PCR. Chemical analysis of the PCR mixture reveals that at the end of PCR there is usually plenty of primers and NTPs so that PCR should continue for further cycles before saturation is observed due to consumption of a limiting reagent. Experimentally, if you double the concentration of the primers, you do not observe twice the PCR product. Thus, the amplicon yield of PCR is usually less than predicted based on the primer concentrations.
What is causing the PCR to saturate prematurely? The answer is that double-stranded DNA is an excellent inhibitor of DNA polymerase. This can be demonstrated experimentally by adding a large quantity of non-extensible “decoy” duplex DNA to a PCR and comparing the result to a PCR without the added duplex. The result clearly shows that the reaction with added duplex DNA shows little or no amplification while the control amplifies normally. The reason why duplex DNA inhibits DNA polymerase is that the polymerase binds to the duplex rather than binding to the small quantity of duplex arising from the primers binding to target strands during the early cycles of PCR.
Application of the Inhibition Principle to Multiplex PCR Design
The concept of amplicon inhibition of PCR is particularly important for multiplex PCR design. Consider a multiplex reaction in which there are plenty of NTPs available. It is expected that if one of the amplicons is produced more efficiently than the others, then it will reach saturation and inhibit the polymerase from subsequently amplifying the other amplicons.
To achieve uniform amplification of the different targets, the primers must be designed to bind with equal efficiency to their respective targets. Binding equally does not mean “matched Tm’s.” This requires the use of accurate thermodynamic parameters (i.e., by not using the older methods for Tm prediction) and also accounting for the effects of competing equilibria, which requires the use of the coupled multi-state equilibrium model described in Subheadings 2.1 and 2.4 as well as the other principles described in this chapter.

